At Thrafford, we’re pioneering the next generation of cell therapy manufacturing — making treatments more affordable, scalable, and accessible to patients everywhere. Our breakthrough technologies are designed to overcome the biggest challenges in the cell therapy industry: cost, complexity, and limited reach.
Our Three-Pillar Approach
AffordabilityShifting from Autologous to Allogeneic Therapies: Today’s autologous (patient-specific) CAR-T therapies are expensive and time-intensive. Thrafford is transforming this model with healthy donor–derived allogeneic cells — one production batch serving multiple patients. This shift dramatically lowers the cost per treatment, paving the way for truly accessible advanced therapies.
 One batch manufacturing. Multiple patients. Lower cost.
Scalability
One batch manufacturing. Multiple patients. Lower cost.
Scalability
Parallel, Modular, and Efficient: Traditional centralized manufacturing struggles to meet growing demand. Thrafford’s modular platform enables parallel, multi-batch processing, boosting production capacity by up to 40× compared to conventional systems. With semi-automation and built-in quality control, we deliver consistent, high-quality results at scale. A single, flexible platform for affordable, high-throughput, and reliable cell therapy manufacturing.
 Scalable design. Consistent quality. 40× more capacity.
Accessibility
Scalable design. Consistent quality. 40× more capacity.
Accessibility
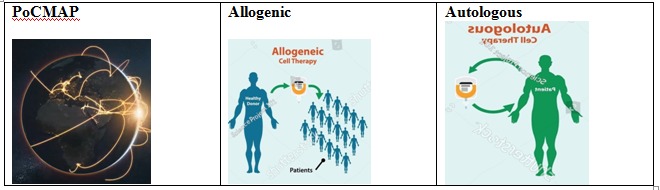
India’s First Decentralized Point-of-Care Manufacturing Platform (PoCMAP): Our patented PoCMAP platform brings CAR-T manufacturing directly into hospital environments. This decentralized, semi-automated system eliminates the need for cryopreservation and long-distance logistics — ensuring therapies reach patients faster, safer, and closer to home.
 Manufacturing at the point of care — where patients need it most.
Manufacturing at the point of care — where patients need it most.
The PoCMAP Advantage
- Decentralized & Scalable: Localized manufacturing at hospitals for real-time therapy delivery.
- Parallel Processing: Simultaneous multi-batch production enhances efficiency and throughput.
- Reagent-Agnostic Flexibility: Compatible with multiple cell types (T, NK, dendritic) and both autologous & allogeneic workflows.
- Integrated Quality Control: Real-time sterility testing and in-process monitoring ensure safety and consistency.
- Reduced Logistics & Time: Fewer freeze–thaw steps and minimal transport reduce delays and risk.
Engineered for the Future of Medicine
Thrafford’s PoCMAP is more than a manufacturing system — it’s a biomanufacturing revolution. By merging semi-automation, modular design, and real-time quality control, we empower hospitals to produce personalized therapies on demand.
Applications- CAR-T cell therapies
- NK cell and dendritic cell therapies
- Autoimmune and infectious diseases
- Solid tumors and in-vivo gene editing (future-ready)
At Thrafford, we’re pioneering the next generation of cell therapy manufacturing — making treatments more affordable, scalable, and accessible to patients everywhere. Our breakthrough technologies are designed to overcome the biggest challenges in the cell therapy industry: cost, complexity, and limited reach.
Our Three-Pillar Approach
Affordability
Shifting from Autologous to Allogeneic Therapies: Today’s autologous (patient-specific) CAR-T therapies are expensive and time-intensive. Thrafford is transforming this model with healthy donor–derived allogeneic cells — one production batch serving multiple patients.
 One batch manufacturing. Multiple patients. Lower cost.
One batch manufacturing. Multiple patients. Lower cost.
Scalability
Parallel, Modular, and Efficient: Traditional centralized manufacturing struggles to meet growing demand. Thrafford’s modular platform enables parallel, multi-batch processing, boosting production capacity by up to 40× compared to conventional systems.
 Scalable design. Consistent quality. 40× more capacity.
Scalable design. Consistent quality. 40× more capacity.
Accessibility
India’s First Decentralized Point-of-Care Manufacturing Platform (PoCMAP): Our patented PoCMAP platform brings CAR-T manufacturing directly into hospital environments, ensuring therapies reach patients faster, safer, and closer to home.
 Manufacturing at the point of care — where patients need it most.
Manufacturing at the point of care — where patients need it most.
The PoCMAP Advantage
- Decentralized & Scalable: Localized manufacturing at hospitals for real-time therapy delivery.
- Parallel Processing: Simultaneous multi-batch production enhances efficiency and throughput.
- Reagent-Agnostic Flexibility: Compatible with multiple cell types (T, NK, dendritic) and both autologous & allogeneic workflows.
- Integrated Quality Control: Real-time sterility testing and in-process monitoring ensure safety and consistency.
- Reduced Logistics & Time: Fewer freeze–thaw steps and minimal transport reduce delays and risk.
Engineered for the Future of Medicine
Thrafford’s PoCMAP is more than a manufacturing system — it’s a biomanufacturing revolution. By merging semi-automation, modular design, and real-time quality control, we empower hospitals to produce personalized therapies on demand.
Applications- CAR-T cell therapies
- NK cell and dendritic cell therapies
- Autoimmune and infectious diseases
- Solid tumors and in-vivo gene editing (future-ready)
